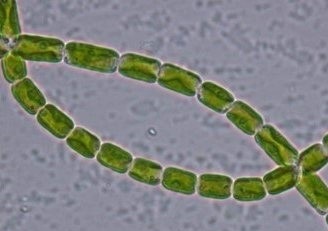
Nutrient removal from vinasse by Klebsormidium flaccidum and biomass production with economic potential
DOI:
https://doi.org/10.20873/jbb.uft.cemaf.v13n1.18757Keywords:
effluent, microalgae, remediation, sustainabilityAbstract
Ethanol is a sustainable alternative for mobility due to low carbon emissions. In Brazil, sugarcane ethanol production generates a large amount of vinasse, and its primary use in fertirrigation causes negative environmental impacts. This study aims to cultivate the microalga Klebsormidium flaccidum using vinasse as a source of nutrients while simultaneously producing biomass of economic value. Three treatments with concentrations of 10%, 20%, and 30% of diluted vinasse were tested, designated as T1, T2, and T3, respectively. A culture using Bold Basal Medium was considered as control. Although the treatments resulted in lower cell densities compared to the control, there was an increase in dry mass (DM) due to nutrient availability. Photosynthetic pigments (chlorophyll a and carotenoids) were lower in the treatments, but proteins (T2 and T3: 8.72 ± 0.11% DM) increased with the addition of vinasse. Carbohydrates were more abundant in the lower concentration of vinasse (T1: 66.39% DM), while the highest lipid content among the treatments was recorded in T1 (5.74% DM). Among the fatty acids, the presence of myristic acid (T3: 164.4 µg g-1 DM) and palmitic acid (T2: 176.4 µg g-1 DM) was highlighted. K. flaccidum demonstrated high efficiency in removing total nitrogen and NH4+ (> 90%) in treatments T2 and T3. The highest removals of total phosphorus, its ionic form PO4-3, and potassium also occurred in these treatments. Our results highlight the potential of K. flaccidum in nutrient removal from agricultural wastewater, promoting sustainable economy.
Key-words: algae, effluent, remediation, sustainability
References
American Public Health Association (APHA). Standard Methods for the Examination for Water and Wastewater. 22th ed. Washington, D.C.: AWWA, WPCF, 2012.
Beuckels A, Smolders E, Muylaert K. Nitrogen availability influences phosphorus removal in microalgae-based wastewater treatment. Water Research, v. 77, p. 98-106, 2015.
https://doi.org/10.1016/j.watres.2015.03.018
Bligh EG, Dyer WJ. A rapid method of total lipid extraction and purification. Canadian Journal of Biochemistry and Physiology, v. 37, p. 911-917, 1959.
https://doi.org/10.1139/o59-099
Budiyono IS, Sumardiono S, Sasongko SB. Production of Spirulina platensis biomass using digested vinasse as culti-vation medium. Trends in Applied Sciences Research, v. 9, p. 93-102, 2014.
https://doi.org/10.3923/tasr.2014.93.102
Calixto CD, da Silva Santana JK, de Lira EB, Sassi PGP, Rosenhaim R, da Costa Sassi CF, da Conceição MM, Sassi R. Biochemical compositions and fatty acid profiles in four species of microalgae cultivated on household sewage and agro-industrial residues. Bioresource Technology, v. 221, p. 438–446, 2016. https://doi.org/10.1016/j.biortech.2016.09.066
Candido C, Bernardo A, Lombardi AT. Optimization and qualitative comparison of two vinasse pre-treatments aiming at microalgae cultivation. Engenharia Sanitária e Ambiental, v. 26, n. 2, p. 359-367, 2021.
https://doi.org/10.1590/S1413-415220190306
Candido C, Lombardi AT. The physiology of Chlorella vul-garis grown in conventional and biodigested treated vinas-ses. Algal Research, v. 30, p. 79-85, 2018.
https://doi.org/10.1016/j.algal.2018.01.005
Caporgno MP, Taleb A, Olkiewicz M, Font J, Pruvost J, Legrand J, Bengoa C. Microalgae cultivation in urban wastewater: nutrient removal and biomass production for biodiesel and methane. Algal Research, v. 10, p. 232–239, 2015.
https://doi.org/10.1016/j.algal.2015.05.011
Christofoletti CA, Escher JP, Correia JE, Marinho JF, Fonta-netti CS. Sugarcane vinasse: Environmental implications of its use. Waste Management, v. 33, n. 12, p. 2752–2761, 2013.
https://doi.org/10.1016/j.wasman.2013.09.005
Conceição GR, da Silva CS, do Vale TO, dos Santos JN, Matos JBTL, de Almeida PF, Chinalia FA. Culture opera-tional strategies for the production of methane and algal oil using ethanol vinasse effluent. Journal of Applied Phycolo-gy, v. 35, n. 5, p. 2135–2149, 2023. https://doi.org/10.1007/s10811-023-03019-7
Da Silva JC, Lombardi AT. Chlorophylls in Microalgae: Occurrence, Distribution, and Biosynthesis. In: Jacob-Lopes E, Queiroz MI, Zepka LQ (eds). Pigments from Microalgae Handbook. Springer Nature, Switzerland, p. 1-18, 2020.
https://doi.org/10.1007/978-3-030-50971-2
Delgado RT, Guarieiro MS, Antunes PW, Cassini ST, Terre-ros HM, Fernandes VO. Effect of nitrogen limitation on growth, biochemical composition, and cell ultrastructure of the microalga Picocystis salinarum. Journal of Applied Phy-cology v. 33, n. 4, p. 2083–2092, 2021.
https://doi.org/10.1007/s10811-021-02462-8
Engin IK, Cekmecelioglu D, Yücel AM, Oktem HA. Evalua-tion of heterotrophic and mixotrophic cultivation of novel Micractinium sp. ME05 on vinasse and its scale up for bio-diesel production. Bioresource Technology, v. 251, p. 128-134, 2018.
https://doi.org/10.1016/j.biortech.2017.12.023
Ferreira G, Fernandes D, Pinto LR, Tasic M, Maciel Filho R. Investigation of Desmodesmus sp. growth in photobioreac-tor using vinasse as a carbon source. Chemical Engineering Transactions, v. 65, p. 721-726, 2018.
https://doi.org/10.3303/CET1865121
Fogg GE, Thake B. Algal Cultures and Phytoplankton Ecolo-gy. The University of Wisconsin Press, London, 175p., 1987.
Franco-Morgado M, Amador-Espejo GG, Pérez-Cortés M, Gutiérrez-Uribe J.A. Microalgae and Cyanobacteria Poly-saccharides: Important Link for Nutrient Recycling and Re-valorization of Agro-Industrial Wastewater. Applied Food Research, v. 3, n. 100296, p. 1-11, 2023. https://doi.org/10.1016/j.afres.2023.100296
Fuess LT, Garcia ML, Zaiat M. Seasonal characterization of sugarcane vinasse: Assessing environmental impacts from fertirrigation and the bioenergy recovery potential through biodigestion. Science of The Total Environment, v. 634, p. 29–40, 2018.
https://doi.org/10.1016/j.scitotenv.2018.03.326
Grobbelaar JU. The microalgal cell with reference to mass cultures. Inorganic algal nutrition. In: Richmond A, Hu Q (eds). Handbook of Microalgal Culture: Biotechnology and Applied Phycology. Wiley-Blackwell, Oxford, p. 123-133, 2013.
https://doi.org/10.1002/9781118567166.ch8
Hori K, Maruyama F, Fujisawa T, Togashi T, Yamamoto N, Seo M et al. Klebsormidium flaccidum genome reveals pri-mary factors for plant terrestrial adaptation. Nature Commu-nications, v. 5, p. 3978, 2014. https://doi.org/10.1038/ncomms4978
Jeffrey SW, Humphrey GF. New spectrophotometric equa-tions for determining chlorophylls a, b, c1 and c2 in higher plants, algae, and natural phytoplankton. Biochemie und Physiologie der Pflanzen, v. 167, p. 191-194, 1975. https://doi.org/10.1016/S0015-3796(17)30778-3
Kadioglu A, Algur OF. Tests of media with vinasse for Chla-mydomonas reinhardtii for possible reduction in vinasse pollution. Bioresource Technology, v. 42, n. 1, p. 1-5, 1992. https://doi.org/10.1016/0960-8524(92)90080-H
Karsten U, Herburger K, Holzinger A. Photosynthetic plastici-ty in the green algal species Klebsormidium flaccidum (Streptophyta) from a terrestrial and a freshwater habitat. Phycologia, v. 56, n. 2, p. 213–220, 2016. https://doi.org/10.2216/16-85.1
Kochert G. Carbohydrate determination by phenol–sulfuric acid method. In: Hellebust JA, Craigie JS (eds). Handbook of Phycological Methods: Physiological and Biochemical Methods. Cambridge University Press, Cambridge, p. 95–97, 1978.
Kumar N, Banerjee C, Chang JS, Shukla P. Valorization of wastewater through microalgae as a prospect for generation of biofuel and high-value products. Journal of Cleaner Pro-duction, v. 362, p. 132114, 2022. https://doi.org/10.1016/j.jclepro.2022.132114
Liu J, Danneels B, Vanormelingen P, Vyverman W. Nutrient removal from horticultural wastewater by benthic filamen-tous algae Klebsormidium sp., Stigeoclonium spp. and their communities: From laboratory flask to outdoor algal turf scrubber (ATS). Water Research, v. 92, p. 61-68, 2016.
https://doi.org/10.1016/j.watres.2016.01.049
Liu J, Pemberton B, Lewis J, Scales PJ, Martin GJO. Wastewater treatment using filamentous algae – A review. Bioresource Technology, v. 298, p. 122556, 2020. https://doi. org/10.1016/j.biortech.2019.122556
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ. Protein measurement with the Folin phenol reagent. Journal of Bio-logical Chemistry, v. 193, n. 265–275, 1951. https://doi.org/10.1016/S0021-9258(19)52451-6
Lyra FH, Carneiro MTWD, Brandão GP, Pessoa HM, Castro EVR. Determination of Na, K, Ca and Mg in biodiesel sam-ples by flame atomic absorption spectrometry (F AAS) us-ing microemulsion as sample preparation. Microchemical Journal, v. 96, n. 1, p. 180-185, 2010. https://doi.org/10.1016/j.microc.2010.03.005
Markou G, Vandamme D, Muylaert K. Microalgal and cyano-bacterial cultivation: The supply of nutrients. Water Rese-arch, v. 65, p. 186–202, 2014. https://doi.org/10.1016/j.watres.2014.07.025
Marques SSI, Nascimento IA, de Almeida PF, Chinalia FA. Growth of Chlorella vulgaris on sugarcane vinasse: The ef-fect of anaerobic digestion pretreatment. Applied Biochemis-try and Biotechnology, v. 171, p. 1933-1943, 2013. https://doi.org/10.1007/s12010-013-0481-y
Mitra D, Van Leeuwen J., Lamsal B. Hetero-trophic/mixotrophic cultivation of oleaginous Chlorella vul-garis on industrial co-products. Algal Research, v. 1, n. 1, p. 40-48, 2012. https://doi.org/10.1016/j.algal.2012.03.002
Montoya T, Gómez J, Mariano M, Jara E, Carrasco N, Tara-zona R et al. Comunidades aereoterrestres de la microalga Klebsormidium (Klebsormidiophyceae, Streptophyta) en costras biológicas del desierto costero peruano. Arnaldoa, v. 26, n. 3, p. 1105-1124, 2019. https://doi.org/10.22497/arnaldoa.263.26317
Oliveira O, Gianesella S, Silva V, Mata T, Caetano N. Lipid and carbohydrate profile of a microalga isolated from was-tewater. Energy Procedia, v. 136, p. 468–473, 2017. https://doi.org/10.1016/j.egypro.2017.10.305
Patel AK, Vadrale AP, Singhania RR, Michaud P, Pandey A, Chen, SJ. et al. Algal polysaccharides: current status and future prospects. Phytochemistry Reviews, v. 22, p. 1167–1196, 2023.
doi:10.1007/s11101-021-09799-5
Ramirez NNV, Farenzena M, Trierweiler JO. Growth of microalgae Scenedesmus sp. in ethanol vinasse. Brazilian Archives of Biology and Technology, v. 57, n. 5, p. 630-635, 2014.
https://doi.org/10.1590/S1516-8913201401791
Renewable Fuels Association (RFA). Annual Ethanol Produc-tion. Washington, D.C.: RFA, 2024. Available at: https://ethanolrfa.org/markets-and-statistics/annual-ethanol-production. Accessed on: Feb. 20, 2024.
Rodrigues DB, Flores EMM, Barin JS, Mercadante AZ, Jacob-Lopes E, Zepka LQ. Production of carotenoids from microalgae cultivated using agroindustrial wastes. Food Re-search International, v. 65, p. 144–148, 2014. https://doi.org/10.1016/j.foodres.2014.06.037
Santana H, Cereijo CR, Teles VC, Nascimento RC, Fernandes MS, Brunale P. et al. Microalgae cultivation in surgacane vinasse: Selection, growth and biochemical characterization. Bioresource Technology, v. 228, p. 133-140, 2017.
https://doi.org/10.1016/j.biortech.2016.12.075
Santos RR, Araújo ODQF, Medeiros JL, Chaloub RM. Culti-vation of Spirulina maxima in medium supplemented with sugarcane vinasse. Bioresource Technology, v. 204, p. 38-48, 2016.
https://doi. org/10.1016/j.biortech.2015.12.077
Sathasivam R, Radhakrishnan R, Hashem A, Abd Allah EF. Microalgae metabolites: A rich source for food and medi-cine. Saudi Journal of Biological Sciences, v. 26, n. 4, p. 709–722, 2019.
https://doi.org/10.1016/j.sjbs.2017.11.003
Silva AS, Griebeler NP, Borges LC. Uso de vinhaça e impac-tos nas propriedades do solo e lençol freático. Revista Brasi-leira de Engenharia Agrícola e Ambiental, v.11, n.1, p. 108-114, 2007.
https://doi.org/10.1590/S1415-43662007000100014
Singh AK, Sharma N, Farooqi H, Abdin MZ, Mock T, Kumar S. Phycoremediation of municipal wastewater by microalgae to produce biofuel. International Journal of Phytoremedia-tion, v. 19, n. 9, p. 805-812, 2017. https://doi.org/10.1080/15226514.2017.1284758
Siqueira JC, Braga MQ, Ázara MS, Garcia KJ, Alencar SNM, Ramos TS, Siniscalchi LAB, Assemany PP, Ensinas AV. Recovery of vinasse with combined microalgae cultivation in a conceptual energy-efficient industrial plant: Analysis of related process considerations. Renewable and Sustainable Energy Reviews, v. 155, p. 111904, 2022. https://doi.org/10.1016/j.rser.2021.111904
Spolaore P, Joannis-Cassan C, Duran E, Isambert A. Com-mercial application of microalgae. Journal of Bioscience and Bioengineering, v. 101, n. 2, p. 87–96, 2006. https://doi.org/10.1263/jbb.101.87
Stein J. Handbook of Phycological methods. Culture methods and growth measurements. Cambridge University Press, 448 p., 1973.
Strickland JDH, Parsons TR. A practical handbook of sea-water analysis. Bulletin of the Fisheries Research Board of Canada, Ottawa, 310p., 1972.
Syaichurrozi I, Jayanudin J. Effect of tofu wastewater addition on the growth and carbohydrate-protein-lipid content of Spirulina platensis. International Journal of Engineering, v. 30, n. 11, p. 1631–1638, 2017. https://doi.org/10.5829/ije.2017.30.11b.02
Tawfik A, Ismail S, Elsayed M, Qyyum MA, Rehan M. Sus-tainable microalgal biomass valorization to bioenergy: key challenges and future perspectives. Chemosphere, v. 296, p. 133812, 2022.
https://doi.org/10.1016/j.chemosphere.2022.133812
Valderrama LT, Del Campo CM, Rodriguez CM, de Bashan LE, Bashan Y. Treatment of recalcitrant wastewater from ethanol and citric acid production using the microalga Chlo-rella vulgaris and the macrophyte Lemna minuscula. Water Research, v. 36, n. 17, p. 4185-4192, 2002. https://doi.org/10.1016/S0043-1354(02)00143-4
Wang Y, Guo W, Yen HW, Ho SH, Lo YC, Cheng CL, Ren N, Chang JS. Cultivation of Chlorella vulgaris JSC-6 with swine wastewater for simultaneous nutrient/COD removal and carbohydrate production. Bioresource Technology, v. 198, p. 619-625, 2015. https://doi.org/10.1016/j.biortech.2015.09.067
Zhang L, Cheng J, Pei H, Pan J, Jiang L, Hou Q et al. Cultiva-tion of microalgae using anaerobically digested effluent from kitchen waste as a nutrient source for biodiesel production. Renewable Energy, v. 115, p. 276-287, 2018. https://doi.org/10.1016/j.renene.2017.08.034
Zheng S, He M, Jiang J, Zou S, Yang W, Zhang Y et al. Effect of kelp waste extracts on the growth and lipid accu-mulation of microalgae. Bioresource Technology, v. 201, p. 80–88, 2016. https://doi.org/10.1016/j.biortech.2015.11.038
Znad H, Al Ketife AMD, Judd S, AlMomani F, Vuthaluru, HB. Bioremediation and nutrient removal from wastewater by Chlorella vulgaris. Ecological Engineering, v. 110, p. 1-7, 2018.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2024 Ronald Tarazona Delgado, Mayara dos Santos Guarieiro, Frederico Pacheco Militão, Valéria de Oliveira Fernandes

This work is licensed under a Creative Commons Attribution 4.0 International License.
Copyright (c) 2024 - Journal of Biotechnology and Biodiversity

This work is licensed under a Creative Commons Attribution 4.0 International License.
Authors who publish with this journal agree to the following terms:
Authors retain copyright and grant the journal right of first publication with the work simultaneously licensed under a Creative Commons Attribution License (CC BY 4.0 at http://creativecommons.org/licenses/by/4.0/) that allows others to share the work with an acknowledgement of the work's authorship and initial publication in this journal.
Authors are able to enter into separate, additional contractual arrangements for the non-exclusive distribution of the journal's published version of the work (e.g., post it to an institutional repository or publish it in a book), with an acknowledgement of its initial publication in this journal.
Authors are permitted and encouraged to post their work online (e.g. in institutional repositories or on their website) prior to and during the submission process, as it can lead to productive exchanges, as well as earlier and greater citation of published work (Available at The Effect of Open Access, at http://opcit.eprints.org/oacitation-biblio.html).


